Topic: Structure of Atom, Test No.: 07, Total MCQs: 10
Congratulations - you have completed Topic: Structure of Atom, Test No.: 07, Total MCQs: 10.
You scored %%SCORE%% out of %%TOTAL%%.
Your performance has been rated as %%RATING%%
Your answers are highlighted below.
Question 1 |
Based on
equation E = –2.178 × 10–18 J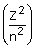 certain conclusions
are written. Which of them is not correct? [NEET 2013]
certain conclusions
are written. Which of them is not correct? [NEET 2013]
Larger the value of n, the larger is the orbit radius | |
Equation can be used to calculate the change in energy when the electron changes orbit | |
For n = 1, the electron has a more negative energy than it does for n = 6 which means that the electron is more loosely bound in the smallest allowed orbit | |
The negative sign in equation simply means that the energy of electron bound to the nucleus is lower than it would be if the electrons were at the infinite distance from the nucleus |
Question 2 |
The
electrons, identified by quantum numbers n and l
(i) n = 4, l = 1, (ii) n = 4, l = 0, (iii) n = 3, l = 2, (iv) n = 3, l = 1
can be placed in order of increasing energy, from the lowest to highest, as:
(i) n = 4, l = 1, (ii) n = 4, l = 0, (iii) n = 3, l = 2, (iv) n = 3, l = 1
can be placed in order of increasing energy, from the lowest to highest, as:
(iv) < (ii) < (iii) < (i) | |
(ii) < (iv) < (i) < (iii) | |
(i) < (iii) < (ii) < (iv) | |
(iii) < (i) < (iv) < (ii) |
Question 3 |
The atomic
number of elements X, Y and Z are 19, 21 and 25 respectively. The number of
electrons present in the 'M' shell of these elements follows the order
Z > X > Y | |
X > Y > Z | |
Z > Y > X | |
Y > Z > X |
Question 4 |
The
de-Broglie wavelength of a tennis ball of mass 60 g moving with a velocity of
10 meters per second is approximately
10–33 m | |
10–31 m | |
10–16 m | |
10–25 m |
Question 5 |
The
electronic configuration of an element is 1s2 2s2 2p6
3s2 3p6 3d5 4s1. This presentation
is
excited state | |
ground state | |
cationic form | |
anionic form |
Question 6 |
The electronic configuration of Cr3+ is
[Ar]3d54s1 | |
[Ar]3d24s1 | |
[Ar]3d34s0
| |
[Ar]3d44s2 |
Question 7 |
The energy of a photon is 3 ´ 10–12 ergs. What is its wavelength in nm?
(h = 6.62 × 10–27 ergs, c = 3 ´ 1010 cm/s)
662 | |
1324 | |
66.2 | |
6.62 |
Question 8 |
The
calculated magnetic moment (in Bohr magneton) of Cu2+ ion is
1.73 | |
0 | |
2.6 | |
3.4 |
Question 9 |
The
atomic number of an element ‘M’ is 26. How many electrons are present in the
M-shell of the element in its M3+ state?
11 | |
15 | |
14 | |
13 |
Question 10 |
The
atomic number of an element is 35. What is the total number of electrons
present in all the P-orbitals of the ground state atom of the element
6 | |
11 | |
17 | |
23 |
Once you are finished, click the button below. Any items you have not completed will be marked incorrect.
There are 10 questions to complete.

